Years ago, a friend said to me, “How do you manage to walk through the world as a physicist? You must constantly look at the world around you and think, Wow, it’s incredible what nature is doing!” No need to say: he was a philosophy student.
Actually, I can walk through the world very well. I question nature relatively rarely. However, I remember having a physics wow moment once when I was a teenager that kept me thinking.
At the time, I was playing around with a coaster (I said it, teenager, with what else?) and frayed the end. Then I held it up close to my face and looked at the edge. Actually, I thought to myself, I should now see a hard, thin line after all. But instead, I saw a flowing progression from coaster-white to wall-green. And I thought: Huh? Why is that? Surely the coaster is either there or not there, it can’t be “a bit” in my vision? (Here the quantum physicist in me already hinted).
Well, it wasn’t exactly rocket science. The phenomenon is called light refraction and results from the wave property of light. But it kept me busy for an amazingly long time and truly fascinated me.
Which food has the most calories?
I think it’s worth it to look at these little phenomena sometimes, to find the wow moments in everyday life and to ask yourself silly questions sometimes.
You guessed it: The other day I had such a stupid question in my head. I was sitting in a café eating an excellent chocolate tartlet. Chocolate, cream, pastry and raspberries in the middle – delicious! The logical thought that came to me as I ate was: what is the food with the most calories? Does that even exist?

Margarine and cod liver oil
Quite naively, I had considered with my cupcake companion: Peanut butter? Butter? Oil? Birthday cake? Sounds like a good approach.
If you’ve ever looked into bodybuilding, or more generally, weight training and muscle building, you may have heard the rule of thumb: 1g of protein or carbs has 4kcal, fat has 9kcal.
In fact, the calorie content could be easily calculated if you knew all the micro (vitamins and minerals) and macro nutrients (protein, carbohydrates, fat) in a food. That means the highest calorie content would be a food with maximum fat content. Approximately, pure fat should give us a good 900kcal per 100g.
However, some fats contain additives. Think of yogurt half-fat margarine. A 250g packet of butter – the favorite unit of inflation watchers – contains just under 200g of fat! Besides water and scarcely 1-2g protein and carbohydrates each. Thus we get a poor 730kcal per 100g. Olive oil however already comes on 91g fat per 100g and 800kcal.
But where is the holy fat-grail? On a peak value of 901kcal per 100g come: Coconut oil, lard and cod liver oil! Nothing wasted here!
A contemplation about cod liver oil
Does anyone actually still use cod liver oil? What do you do with it? I get a strong David Copperfield vibe (the orphan, not the magician), of a sickly child being nurtured with cod liver oil. Where exactly this image comes from I can’t explain.
Cod liver oil is a brown liquid extracted from the liver of fish (not the fish oil of whales, as some might think). In 1824, scientists discovered health-promoting properties of cod liver oil, for example, against rickets, which is caused by vitamin D deficiency, and cod liver oil has high vitamin A and D content. Whether I would reach for fish-liver juice today in case of vitamin D deficiency, however, I may doubt. But enough about cod liver oil.

Joule: From beer to physics
Is that it? 901kcal per 100g? Not bad, but I think there’s still room for improvement. Let’s consider what calories actually are: a unit of energy. The usual unit in physics is the joule.
One joule is the energy I have to expend to lift a bar of chocolate by one meter.
James Prescott Joule, by the way, was an… interesting man. He lived in the 19th century and was actually a beer brewer, just like his family before him. Some think it was the brewery that first motivated him to look into physics and chemistry to improve operations. He is said to have been very experimental, and to have blown off his eyebrows, flown kites in thunderstorms, and electrocuted a maid into unconsciousness. Whether this was related to increased beer consumption has not been handed down.
What is a calorie?
If you look at the calorie information on a bar of chocolate, you will see that it also indicates the energy content in joules. Actually, the standard for indicating the calorific value of a food should have been changed from calories to joules a long time ago, but what does 2200kJ really tell you?
The problem with calories is that they are not clearly defined. In theory, the definition sounds quite logical:
One calorie is the amount of heat needed to warm a kilogram of water by 1°C.
The problem is that this amount of heat depends on many factors: air pressure, temperature, composition of the water, … A calorie on Mount Everest is much smaller than a calorie on the ground. But of course, that doesn’t change how much flab I put on after eating a bar of chocolate. The so-called thermochemical kilocalorie (so actually 1000 calories), which is relevant for how our body deals with energy, has therefore been defined as: 1kcal = 4184J. Exactly.
How do you measure calories?
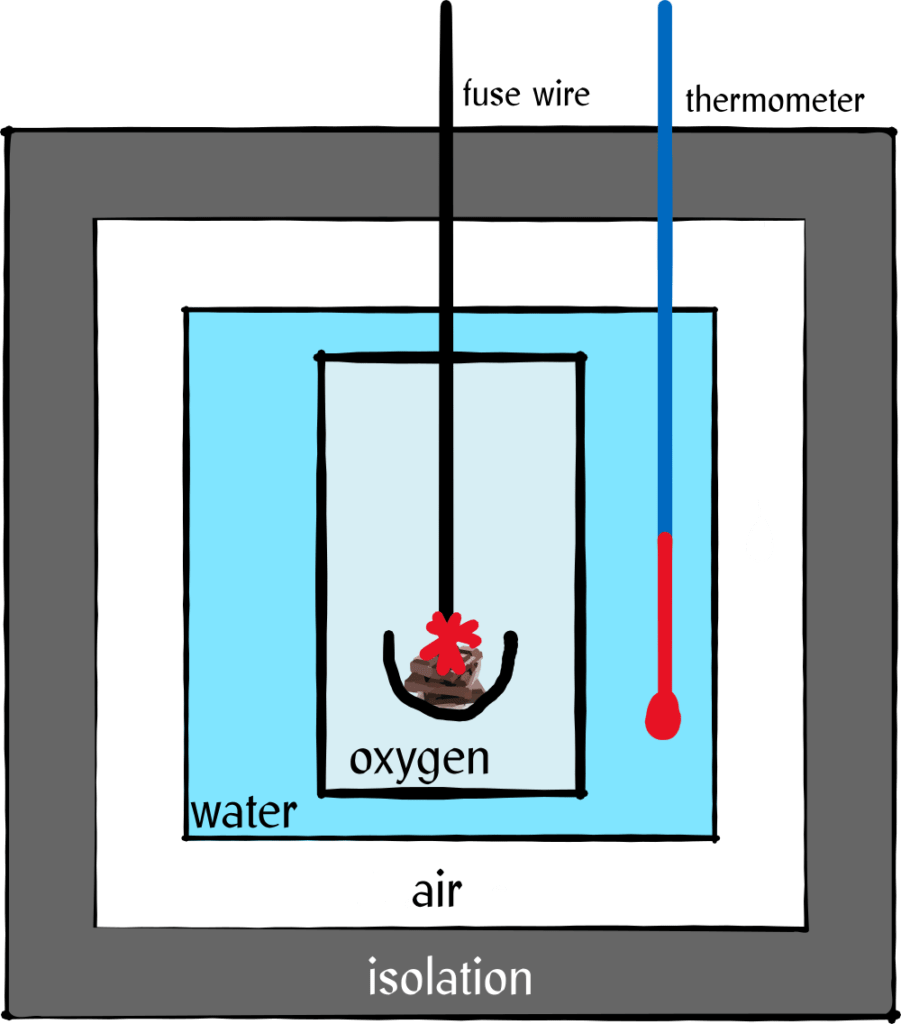
How do food manufacturers find out how many calories are in a product? Test eaters? Doesn’t sound like the worst job in the world. But no, they use a bomb calorie meter and that’s about as cool as it sounds!
To do this, the researchers put a certain amount of the substance for which they want to determine the calorific value into a steel container and blast it away! Of course, this happens under very scientific conditions, the container is filled with oxygen gas, there is well-tempered water around it, the whole thing is isolated and so on, but finally they ignite the sample and blow it up. After that, they measure how much the water has heated up, that is, how much energy has been released in the process.
The problem with this method is that the body cannot extract all of its energy from a substance. Toilet paper burns pretty well, but it’s not particularly nutritious. The so-called physiological calorific value that is written on the food is therefore smaller than the physical one that we get by blasting it away.
How many calories does a black hole have?
But we are in physics here and someone else can tell you much better about digestion and metabolism. I’m more interested in the physical calorific value, that is, the pure energy content of food or objects. How can I beat the calorie content of cod liver oil?
Einstein taught us: energy and mass are equivalent. When mass is converted into energy, quite a bit of energy comes out. Our sun has been doing this for 4 billion years and is far from being exhausted. So the object with the highest energy density must be the object with the highest mass density and that is – you guessed it – a black hole!

Calculating the calorie content is actually quite simple. We use Einstein’s famous equation ![]() , where
, where ![]() is the mass, 100g, and
is the mass, 100g, and ![]() is the speed of light, just under 300 million meters per second. That is
is the speed of light, just under 300 million meters per second. That is ![]() Joules or:
Joules or:
2 Trillion Kilocalories
Calorific value of 100g black hole
That’s 240 million liters of cod liver oil (our new standard energy unit for calorific value). A black hole with 100g mass would have a radius of about ![]() m. A proton has a radius of
m. A proton has a radius of ![]() m – so it is… much larger. Our 100g black hole relates to a proton like a proton relates to a sesame seed.
m – so it is… much larger. Our 100g black hole relates to a proton like a proton relates to a sesame seed.
It is not impossible that black holes this small can exist. It is estimated that black holes can become as small as the Planck length – the fundamentally smallest length possible – but not smaller because quantum effects dominate at smaller lengths (and you thought I couldn’t get the hang of quantum physics). Thus the smallest imaginable black hole would have a mass of only 22 micrograms. This would have almost 470 000 kcal – as much as 52 liters of cod liver oil!
Do you like what you read? Then you can buy me a coffee here! And if you don’t want to miss any new posts, don’t forget to subscribe to my blog.
